Cancer Immunotherapy: What is CD47 blockade?
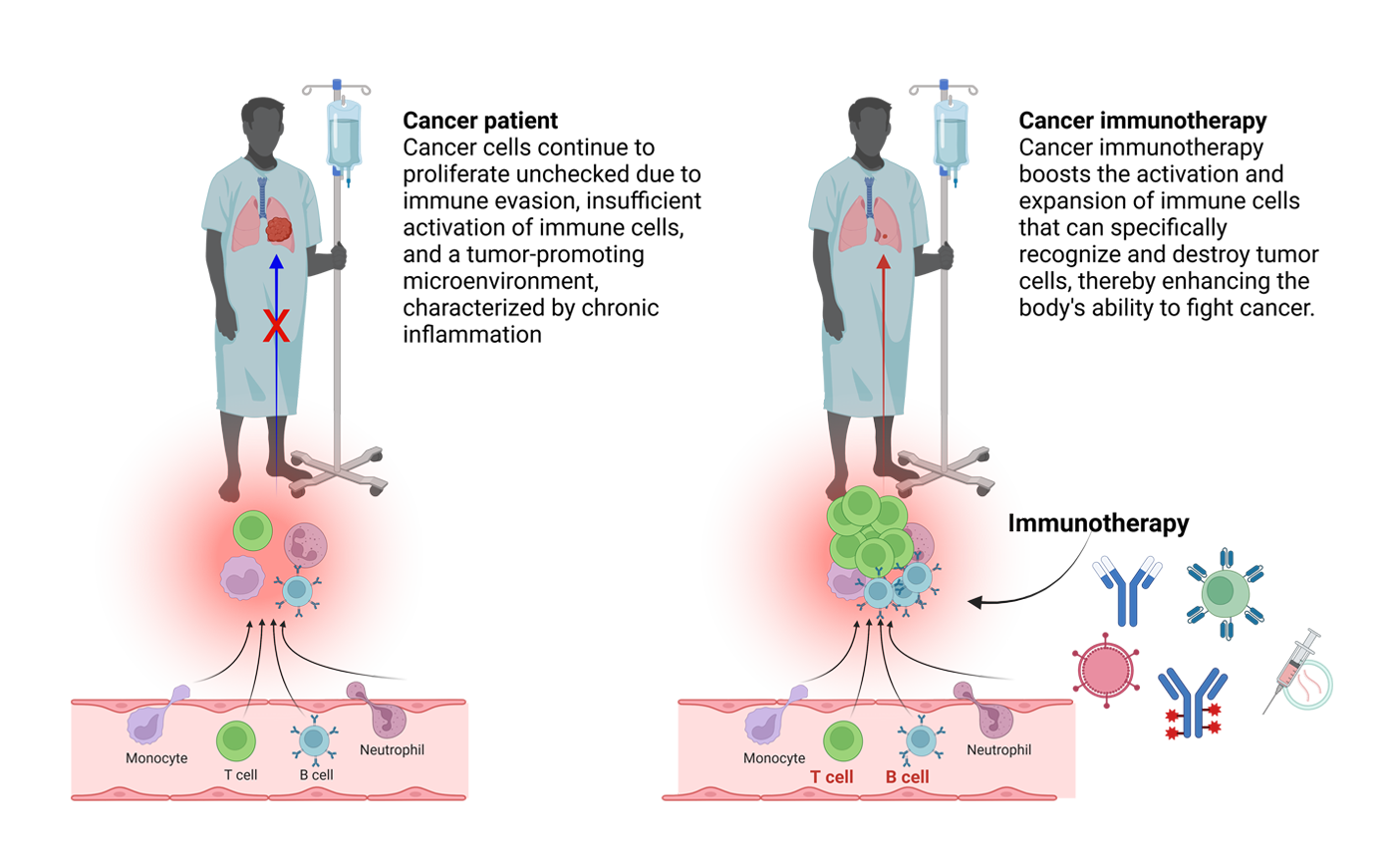
Cancer immunotherapy is a treatment strategy that harnesses the body’s own immune system to identify and eliminate cancer cells. Unlike conventional therapies, such as chemotherapy and radiation that directly target tumor cells, immunotherapy aims to re-educate or boost the immune response to recognize cancer as foreign. This approach encompasses various modalities including checkpoint inhibitors, cancer vaccines, and adoptive cell therapies. Checkpoint inhibitors, for instance, work by blocking proteins that suppress T‑cell activity, thereby restoring the immune system’s ability to attack cancer cells. The underlying principle is to shift the balance in favor of the immune system, enabling it to continuously surveil, recognize, and eradicate malignant cells. This dynamic, patient‑tailored treatment paradigm represents a significant evolution in oncology, aiming not only to shrink tumors but also to induce durable, long‑term remission.
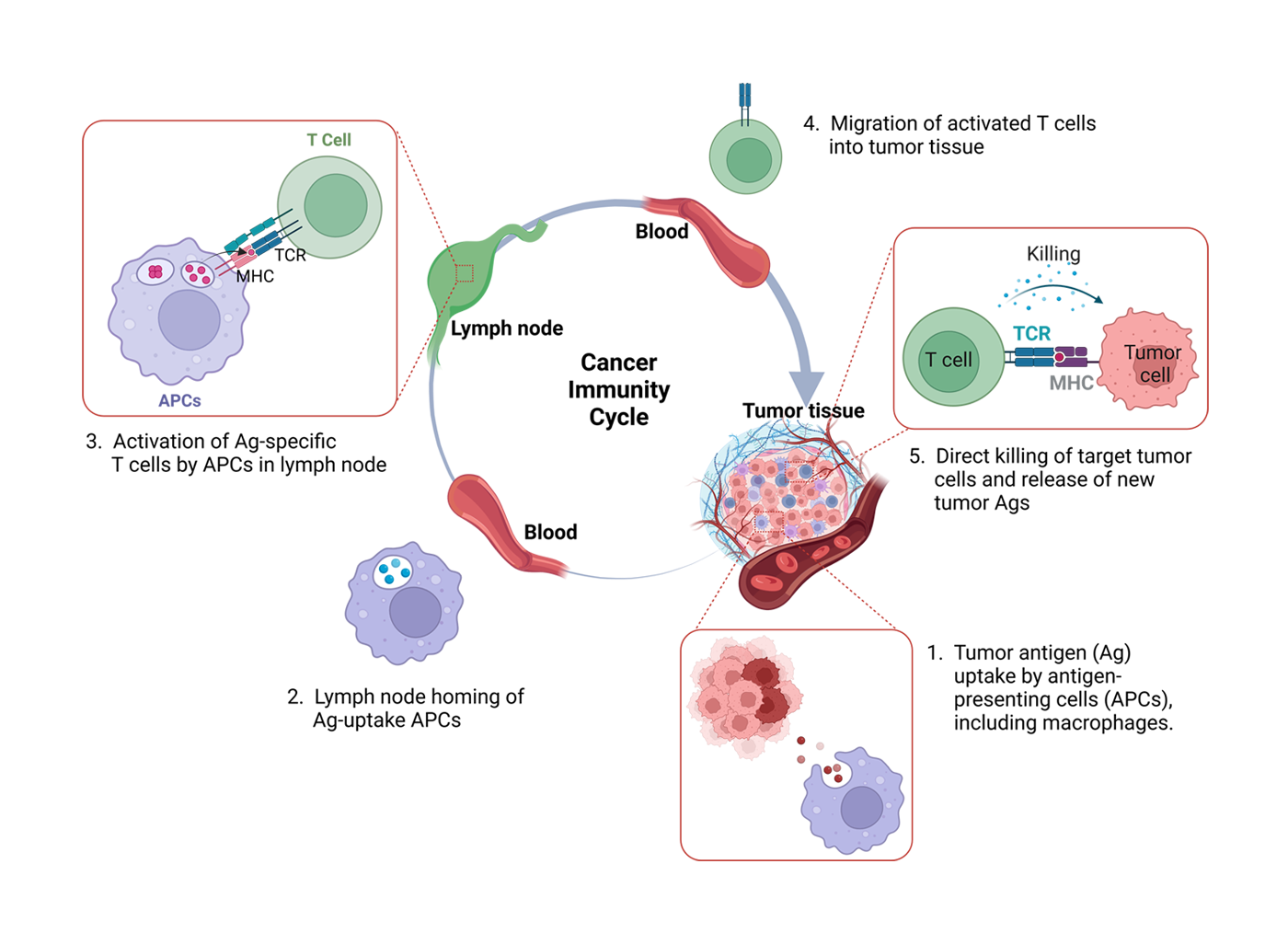
The cancer immunity cycle outlines the complex, multi‑step process by which the immune system combats cancer. It begins with the release of tumor antigens from dying cancer cells, which are then captured by antigen‑presenting cells (APCs) such as macrophages and dendritic cells. These APCs process the antigens and migrate to lymph nodes, where they present the information to T cells, leading to T cell activation. Activated T cells then travel through the bloodstream to the tumor site, where they recognize and bind to cancer cells expressing these antigens. Once in contact, the T cells induce the destruction of cancer cells, which in turn releases additional tumor antigens and perpetuates the cycle.
All cancer immunotherapies work by reinforcing and accelerating this cycle, yet they do not always lead to complete eradication of cancer on their own. However, if a specific immunotherapy can empower patients to sustain the cancer immunity cycle even after the direct effects of the treatment diminish, the likelihood of achieving a true cure increases dramatically. From this perspective, there is a growing need for combination strategies that integrate immunotherapies with different mechanisms. By working in harmony, these therapies can more effectively bolster and maintain the cancer immunity cycle, paving the way for durable, long‑term control—or even complete remission—of the disease.
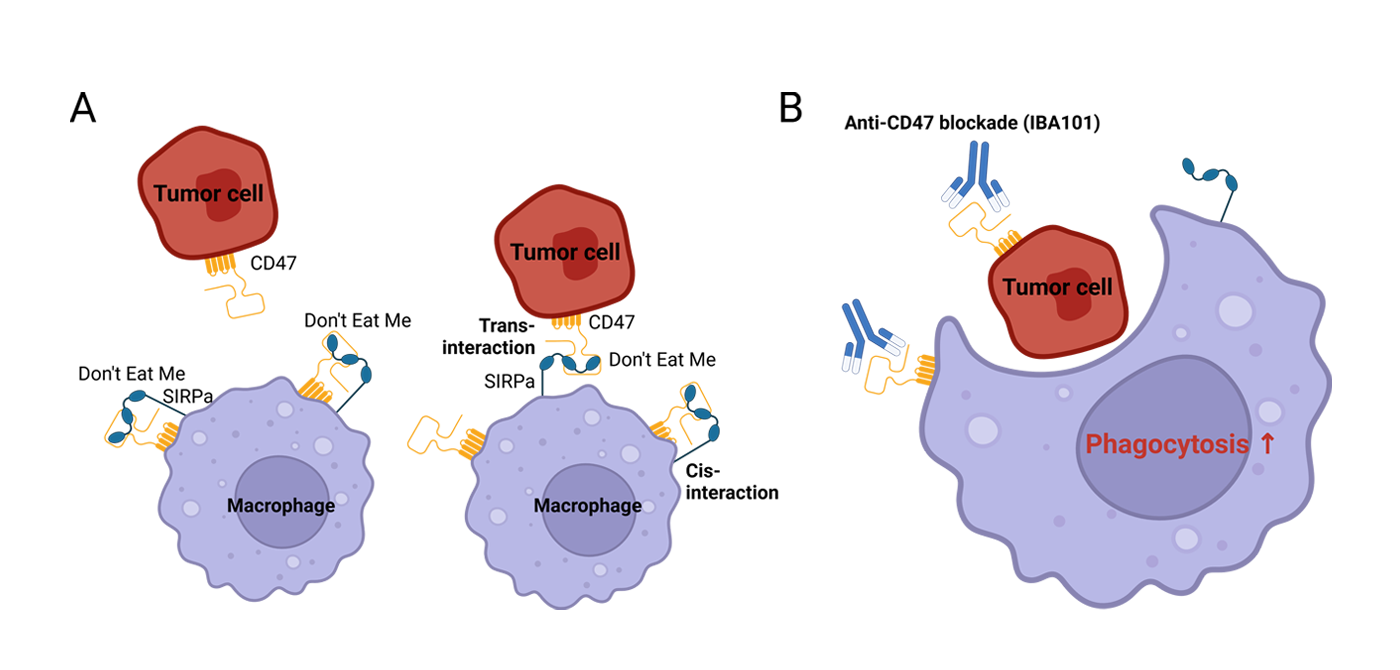
Tumor cells often evade immune destruction by expressing signals that inhibit their clearance by the body’s phagocytic cells. One key mechanism involves the "Don't Eat Me" signal, primarily mediated by the protein CD47. Expressed on the surface of many cancer cells, CD47 binds to signal regulatory protein alpha (SIRPα) on macrophages, effectively sending a message to avoid phagocytosis. This interaction serves as a crucial immune checkpoint, allowing cancer cells to escape detection and destruction by the innate immune system. CD47 blockade, therefore, represents a novel therapeutic approach that targets this immune evasion strategy. By inhibiting the CD47-SIRPα interaction, CD47 blockade therapies can re-engage macrophages, prompting them to recognize and engulf tumor cells. This not only disrupts the protective shield that cancer cells rely on but also promotes a broader immune response that can enhance the efficacy of combination treatments. Such strategies are at the forefront of developing next‑generation immunotherapies, particularly for solid tumors.
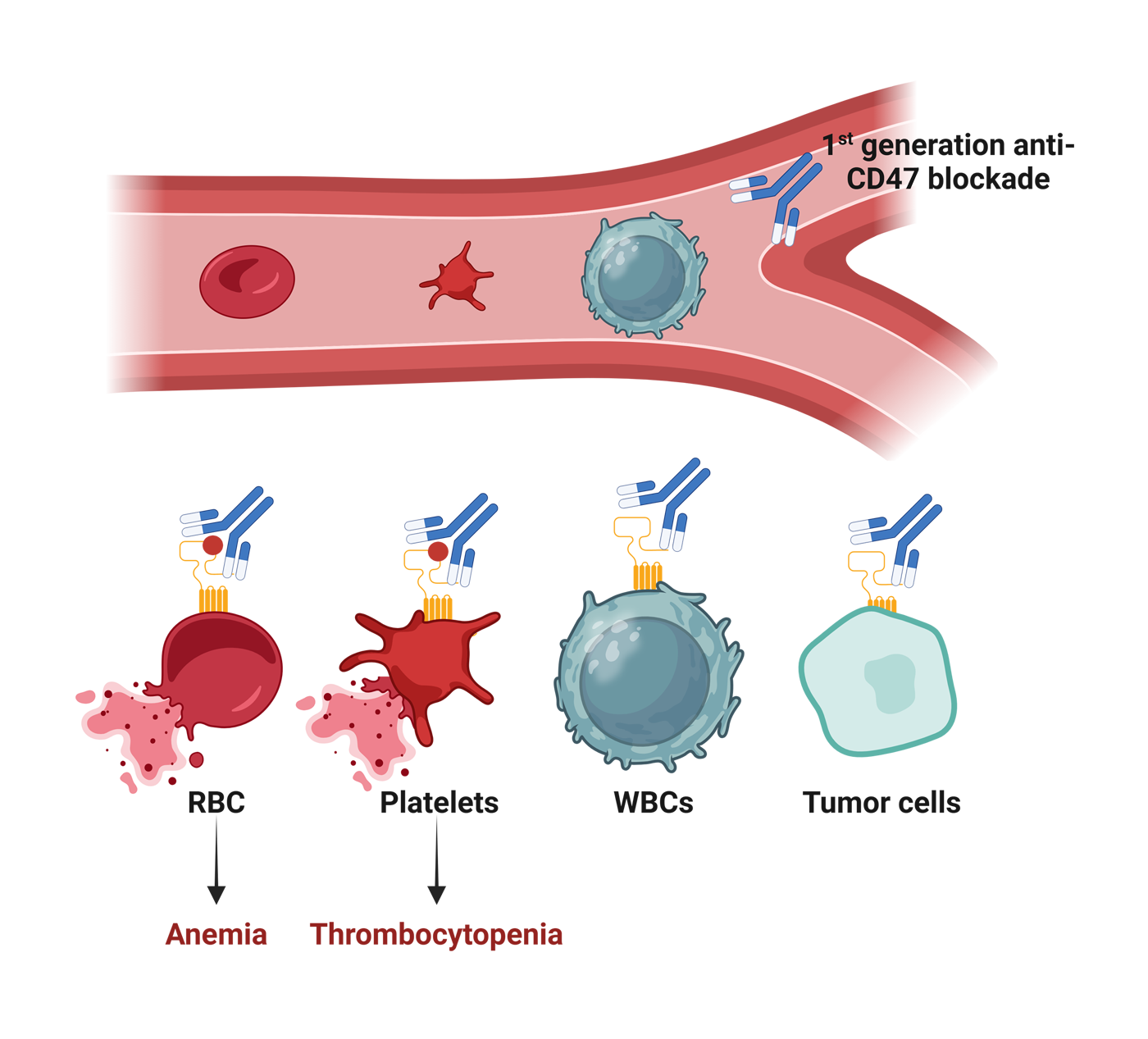
Traditional CD47-blocking monoclonal antibodies (mAbs) have attracted significant attention as immuno-oncology targets due to their ability to enhance innate immunity and inhibit critical immune evasion mechanisms of cancer cells. However, these antibodies bind strongly to red blood cells (RBCs) and platelets, triggering cell death. This off-target binding results in severe anemia and thrombocytopenia in cancer patients, posing major safety concerns during clinical development. Consequently, the development of traditional CD47-blocking mAbs was gradually discontinued. The challenges observed with these first-generation agents underscore the need for next-generation strategies that can safely harness the benefits of CD47 blockade without compromising normal cell function.
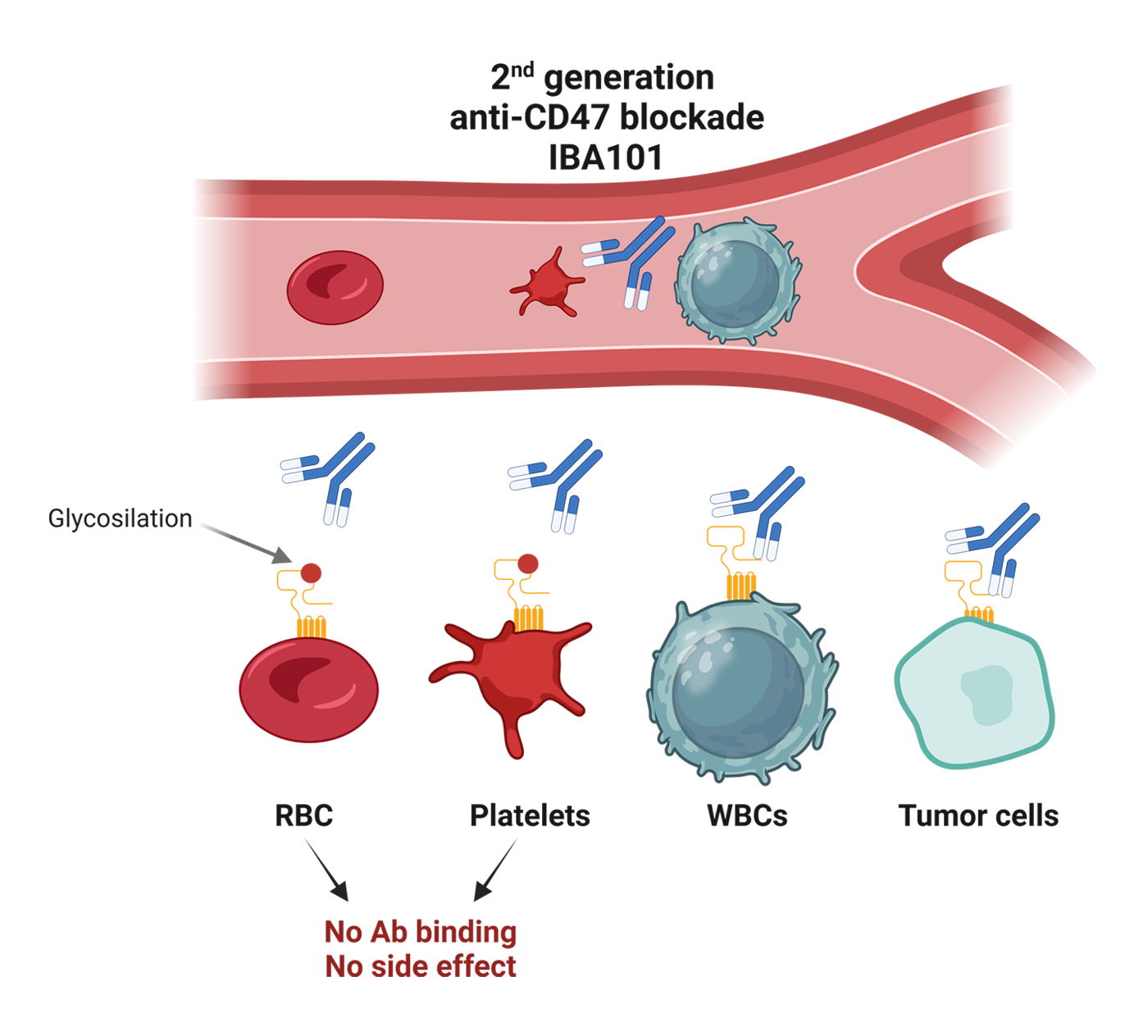
IBA101 represents a breakthrough in CD47 blockade technology. IBA101 is a humanized anti-human CD47 monoclonal antibody with a human IgG4 Fc, designed to preferentially bind nucleated cells—such as immune and tumor cells—while sparing anucleate cells like red blood cells and platelets. IBA101 effectively promotes macrophage-mediated phagocytosis of tumor cells. In a humanized animal tumor model, although IBA101 alone shows modest tumor suppression, its combination with anti-PD1 monoclonal antibody results in complete tumor growth inhibition and regression, all without notable damage to major organs. Moreover, IBA101 has demonstrated synergistic anti-tumor effects not only with PD-1/PD-L1 blockade but also with agents such as TIGIT antagonists and 4‑1BB agonists. Given its ability to efficiently convey tumor antigen information from cancer cells to T cells, IBA101 is well positioned to work synergistically with a variety of immunotherapeutic agents, potentially enhancing overall treatment efficacy and expanding therapeutic options for patients.

